Where is ultraviolet radiation located?

Ultraviolet light is a form of electromagnetic radiation that is responsible for summer tans as well as sunburns. It is also the source of the glow that is produced by black-light posters. However, living tissue may be severely damaged by excessive exposure to UV light. The sun is the source of electromagnetic radiation, which may travel across space as waves or particles, traveling at a variety of wavelengths and frequencies. The term “electromagnetic spectrum” (EM spectrum) refers to this comprehensive range of wavelengths. In general, the spectrum is split up into seven different areas based on the decreasing wavelength and rising energy and frequency in each successive zone. Radio waves, microwaves, infrared (IR), visible light, ultraviolet (UV), X-rays, and gamma rays are the common names for the many types of electromagnetic radiation. To make the most use of this knowledge, we must know more about where ultraviolet radiation is located. Follow us.
- UVA, or very close to UV (315–400 nm),
- UVB, or intermediate UV (280–315 nm),
- UVC, or far UV (180–280 nm),

The following is an excerpt from the guide: “Radiations whose wavelengths range from 10 nm to 180 nm are commonly referred to as vacuum or severe UV.” These wavelengths can only be sent in a vacuum because air interferes with their transmission. Within the electromagnetic spectrum, ultraviolet radiation is located in the area between visible light and X-rays. It has frequencies ranging from around 8 x 1014 to 3 x 1016 cycles per second, often known as hertz (Hz), with wavelengths ranging from approximately 380 nanometers (1.5 105 inches) to approximately 10 nm (4 107 inches). The United States Navy’s “Ultraviolet Radiation Guide” says that ultraviolet light is made up of three different sub-bands:
Related Article: What UV light is best for plants
Ionization
UV light has a sufficient amount of energy to sever chemical bonds. Ionization is a process that occurs when electrons separate from their associated atoms and may be caused by UV photons because of the greater energy that they possess. The vacancy that is created as a consequence of this process has an effect on the chemical characteristics of the atoms, causing them to either establish or break chemical bonds when they normally would not.
The processing of chemicals may benefit from this, but it also has the potential to be harmful to materials and biological tissue. This damage can be beneficial, for example in disinfecting surfaces, but it can also be harmful, particularly to skin and eyes, which are most adversely affected by higher-energy UVB and UVC radiation. This damage can also be beneficial in the sense that it can be beneficial in disinfecting surfaces.

UV repercussions
The sun is the primary source of ultraviolet radiation that humans are exposed to in the natural environment. According to the National Toxicology Program, ultraviolet radiation makes up just about ten percent of sunshine, and of that, only about a third of it is able to get through the atmosphere and reach the ground (NTP). The amount of UVA radiation that is produced by the sun and travels all the way to the equator is 95 percent, whereas the amount of UVB radiation is just 5 percent.
Due to the fact that ozone, molecular oxygen, and water vapor in the high atmosphere totally absorb the shortest UV wavelengths, solar energy does not transmit any UVC that is quantifiable to the surface of the Earth. According to the National Toxicology Program’s “13th Report on Carcinogens,” however, “broad-spectrum ultraviolet radiation [UVA and UVB] is the strongest and most destructive to living beings.”
Related Article: Is UV-C light harmful for air ducts and electronics?
Sunburn
A tan is the skin’s response to being exposed to the sun’s potentially cancer-causing UVB radiation. A tan is essentially the outcome of the body’s natural defense system being activated after being exposed to sunlight. This is made up of a substance known as melanin, which is a pigment that is generated by cells in the skin known as melanocytes. Melanin is responsible for absorbing ultraviolet light and then releasing it as heat. When the body detects that it has been damaged by the sun, it releases melanin into the cells that are nearby in an effort to shield those cells from further deterioration. The pigment is responsible for the darkening of the skin.
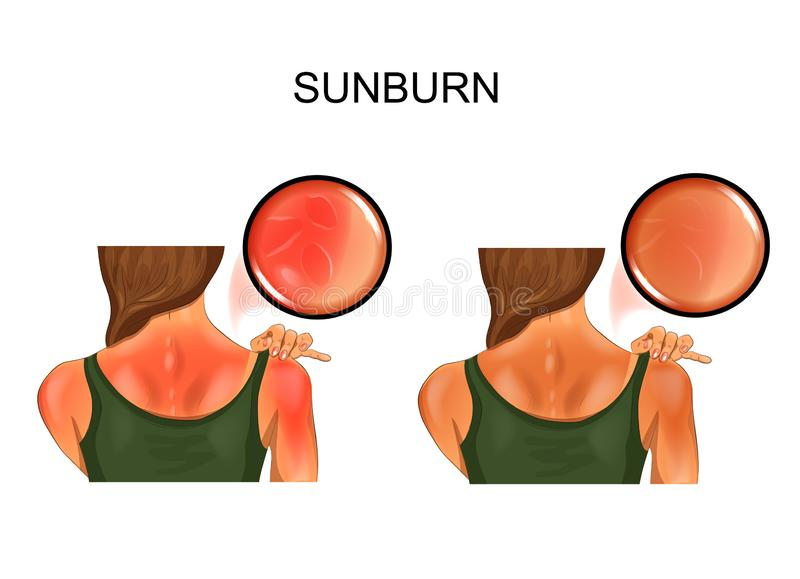
In an interview conducted in 2013, Gary Chuang, an associate professor of dermatology at Tufts University School of Medicine, said that melanin acts as a natural sunscreen. However, prolonged exposure to UV radiation may cause the body’s defensive mechanisms to become overwhelmed. When this occurs, a poisonous reaction takes place, which leads to a sunburn. UV rays have the potential to cause harm to the body’s DNA.
When the body detects damage of this kind, it rushes blood to the affected region in order to speed up the recovery process. Additionally, there is a painful inflammation that develops. After overexposure to the sun for more than half a day, the telltale red lobster appearance of a sunburn will often begin to become seen and felt on the affected area of the skin.
Sometimes the cells whose DNA has been changed by the sun’s rays transform into problematic cells that do not die but instead continue to multiply and become cancerous. According to Chuang, “UV radiation creates random damage in the DNA and in the process of repairing the DNA,” and as a result, “cells gain the capacity to escape death.” The disease that develops as a consequence is skin cancer, which is the kind of cancer that occurs most often in the United States.
People who consistently get burnt are at a muchincreased risk. Skin Cancer Foundation research shows that a person’s chance of getting melanoma, the most dangerous type of skin cancer, goes up by a factor of two if they have had five or more sunburns in their lifetime.
Related Article: Learning to use and set up Surya Professional disinfectant with UV-C sterilizer box
Additional UV sources
A variety of man-made sources have been developed for the purpose of generating ultraviolet radiation. The Health Physics Society states that “artificial sources include tanning booths, black lights, curing lamps, germicidal lamps, mercury vapor lamps, halogen lights, high-intensity discharge lamps, fluorescent and incandescent sources, and some types of lasers.” Other artificial sources include halogen lights, high-intensity discharge lamps, and germicidal lamps.
An electric current may be sent through vaporized mercury or another gas to produce ultraviolet light, which is one of the most frequent ways UV light is produced. This particular kind of light is often used in tanning booths in addition to being employed for the disinfection of various surfaces. Black lights, which are another use for the lamps, are used to make fluorescent paints and dyes shine. In addition to light-emitting diodes (LEDs), lasers, and arc lamps, different light-emitting diodes (LEDs), lasers, and arc lamps are also available as UV sources for use in industrial, medical, and research settings.
Fluorescence
UV light may be absorbed by a wide variety of things, some of which include minerals, plants, fungi, and microorganisms; other compounds include organic and inorganic molecules. The process of absorption raises the energy level of the material’s electrons to a higher state than they were previously at.
These electrons may then drop back down to a lower energy level by taking a series of smaller steps, at which point they will release some of the energy that they have taken up as visible light. Fluorescent materials, such as those used as pigments in paint or dye, look brighter in sunshine due to the fact that these materials absorb invisible UV radiation and re-emit it at visible wavelengths. As a result of this, they are often used for the production of signage, safety vests, and a variety of other applications where great visibility is essential.
The fluorescence of some minerals and organic molecules may be used to both find and identify the substance in question. Researchers are able to identify certain components of complex biomolecular assemblies, such as living cells, with exceptional sensitivity and selectivity thanks to fluorescent probes, as stated by Thermo Fisher Scientific and Life Technologies.
According to research conducted by the University of Nebraska, fluorescent tubes used for lighting produce ultraviolet radiation with a wavelength of 254 nm in addition to the blue light that is produced when an electric current is passed through mercury vapor. These tubes are used in a variety of lighting applications. “Although not visible, this ultraviolet radiation has a higher energy content than the light that is visible to the human eye. The energy from the ultraviolet light is taken up by the fluorescent coating on the interior of the fluorescent bulb, and it then reemits itself as visible light.
” Similar tubes that do not have the same fluorescent coating as others produce UV light. Due to the ionizing properties of UV radiation, which can kill most microorganisms, this light can be used to clean surfaces. Long-wave UVA light is often produced by black-light tubes by the use of mercury vapor. This kind of light is responsible for making certain dyes and pigments glow.
The fluorescent glow will seem to be more prominent due to the coating on the glass tube, which is a dark-purple filter material. This coating blocks the majority of the visible light. Filtration is not required for uses like disinfection. You may skip that step.
UV astronomy
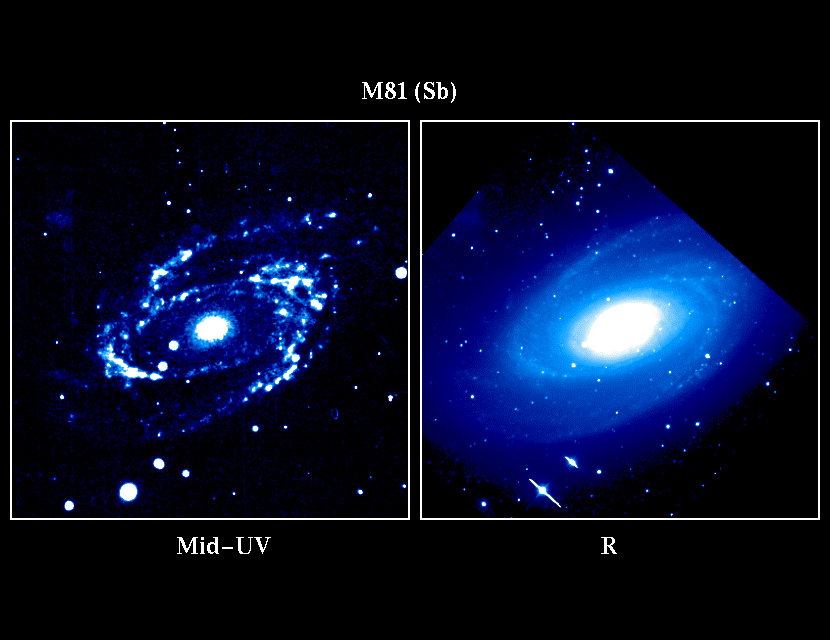
There are a great many more sources of ultraviolet radiation in the cosmos besides the sun. According to NASA, very massive and young stars emit the majority of their light in the ultraviolet wavelength range. Because the Earth’s atmosphere blocks a lot of this UV radiation, especially at shorter wavelengths, observations in the UV part of the electromagnetic spectrum are done with high-altitude balloons and telescopes in orbit that have special imaging sensors and filters.
As stated by Robert Patterson, an astronomy professor at Missouri State University, charge-coupled devices (CCDs) are detectors that are designed to be sensitive to short-wavelength photons. These detectors are used for the majority of the observations that are carried out. Through these studies, it is possible to find out how hot the surfaces of the hottest stars are and if there are gas clouds between the Earth and quasars.
The treatment of cancer
According to Cancer Research UK, there are certain skin disorders that can be treated with UV light (opens in a new tab), despite the fact that prolonged exposure to UV light may cause skin cancer. Psoralen ultraviolet light therapy, often known as PUVA, is a process in which patients either apply a lotion to their skin or take a medicine to make their skin more sensitive to light. After that, ultraviolet (UV) light is directed at the skin.
Vitiligo, eczema, and psoriasis are among the conditions that may be treated with PUVA. It may appear counterintuitive to treat skin cancer with the same thing that caused it, but PUVA may be beneficial because of the effect that UV light has on the production of skin cells. It inhibits growth, which is one of the primary contributors to the progression of the illness.
What was it that gave rise to life?
Recent studies have shown that ultraviolet (UV) radiation may have been an important factor in the development of life on Earth, particularly in the development of RNA. In an article published in 2017 in the Astrophysics Journal, the authors of the study note that it’s possible that red dwarf stars don’t emit enough UV light to kickstart the biological processes that are necessary for the formation of ribonucleic acid, which is required for all forms of life on Earth. This finding was made in light of the fact that the study was conducted in 2017. According to the findings of the research, this discovery may also be of use in the hunt for life in other parts of the cosmos.
